
Representation of the components of the CTD. The nonclinical components... | Download Scientific Diagram

TuraSkills shares tip for writing #Module 2.7 #Sections of clinical summary #Clinical Summary #CTD Summary #Common Technical… | Study skills, Writing tips, Writing

Filing an IND and Beyond: Development of CTD Section 2.6.4, Pharmacokinetics Written Summary Video | Technology Networks
Between Standardisation and Flexibility – Defining Granularity of the eCTD Module 3.2.S for Different Types of Drug Substan

Nonclinical Information in the Common Technical Document: Opportunities for Content Reuse Peggy Zorn, MPI Research Susan Mattano, Pfizer, Inc. - ppt download
PREPARATION AND REVIEW OF CHEMISTRY, MANUFACTURING AND CONTROL (CMC) SECTIONS OF CTD DOSSIER FOR MARKETING AUTHORIZATION Dhruvi

The Challenge of CTD Submissions and Responding to Questions from the Authorities - Trilogy Writing & Consulting GmbH

Figure 1 from Chemistry, Manufacturing and Control (CMC) Evaluations of ANDA Submission in the USA | Semantic Scholar

Map of the study area. The black lines correspond to the CTD sections... | Download Scientific Diagram
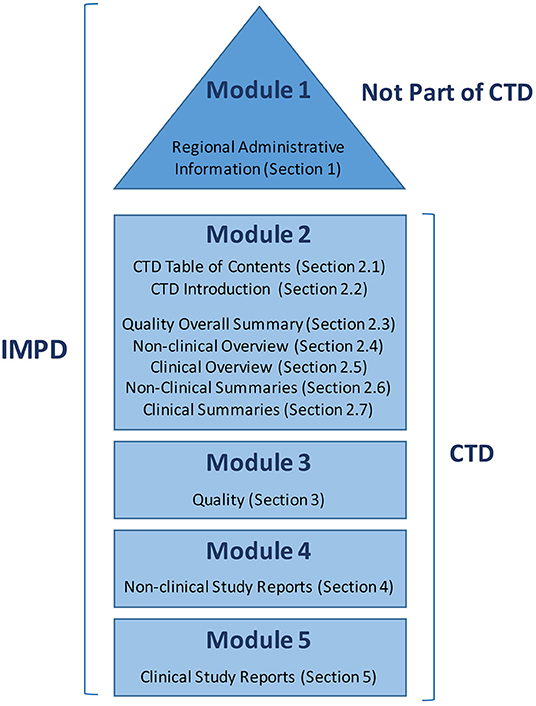




(1).png)








