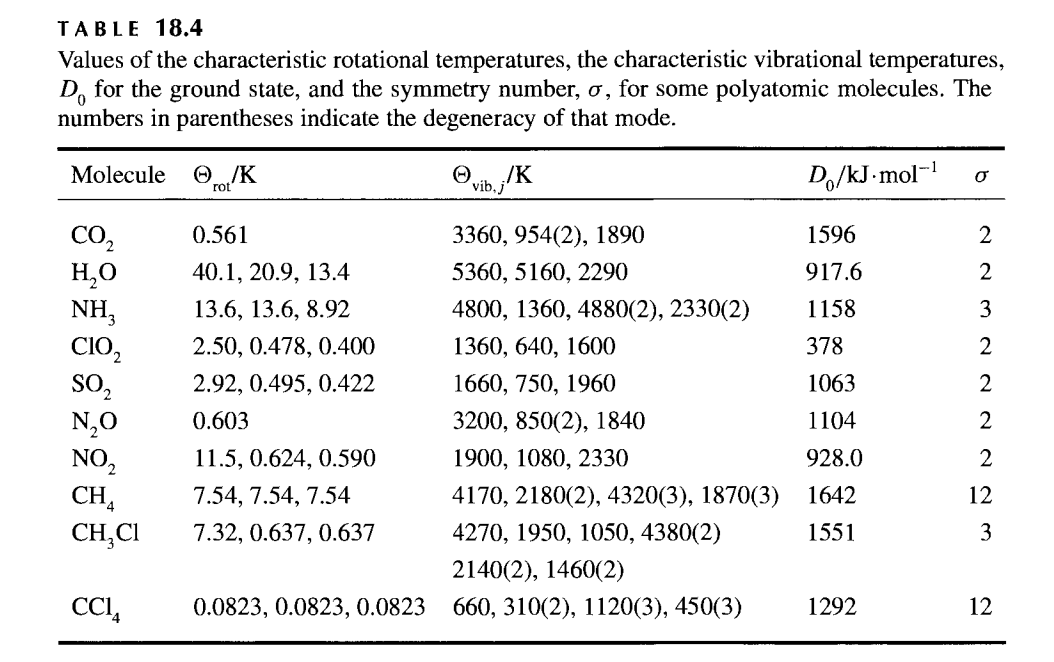
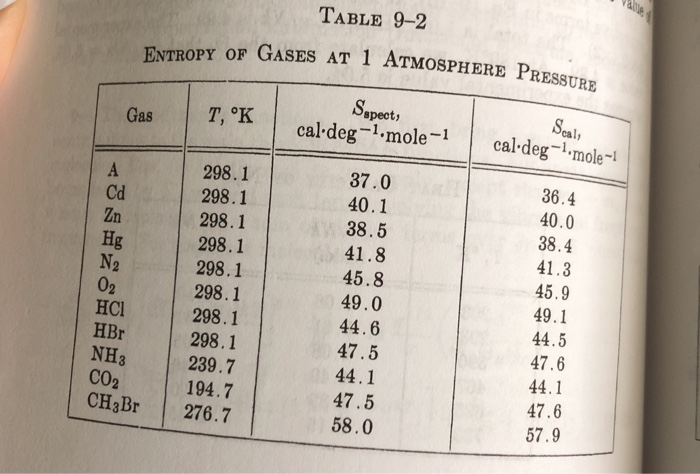
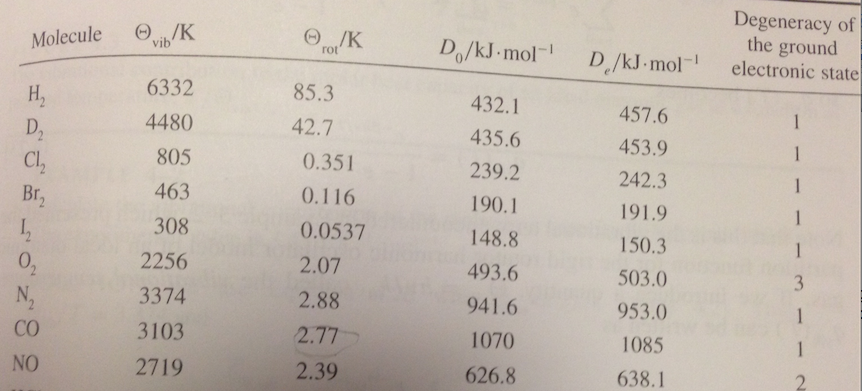SOLVED: The rotational and vibrational partition functions both make use of characteristic temperatures to simplify the expression of the partition function and also to help determine the validity of the expressions (valid

Homework Solution Consider two energy levels at i = 2.05 x 10-21J and j = 2.64 x 10-21J. The occupation numbers of these in a particular system at. - ppt download

The temperature dependence of the characteristic vibrational relaxation... | Download Scientific Diagram
The temperature dependence of the characteristic vibrational relaxation... | Download Scientific Diagram

Temperature effects on the vibration characteristics of a functionally graded thick beam - ScienceDirect

SOLVED: '4-8. Calculale the characteristic vibrational temperature Oub for H,(g) and Dz(g) (, 4401 cm-! and 9 =3112 cm From the definition of O,b' we can write 0 hcv/kg: Then (6.626 x
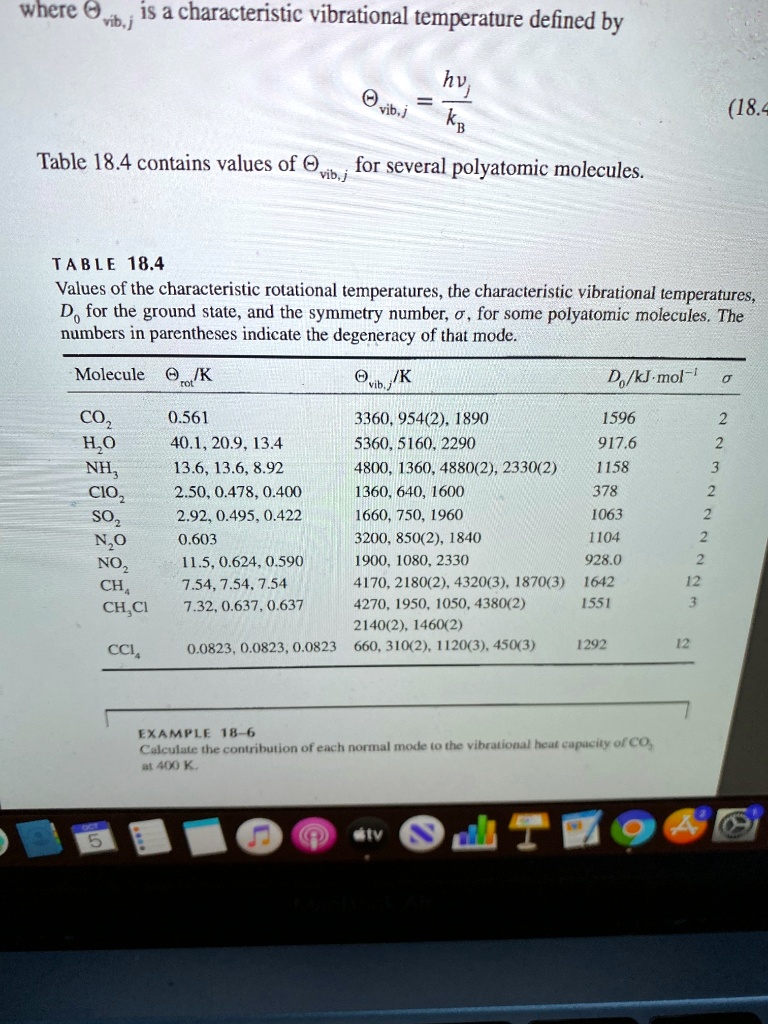
SOLVED: where 0 Mb | iS characteristic vibrational temperature defined by hvi vib j kB (18 = Table 18.4 contains values of vib,j for several polyatomic molecules. TA BLE 18.4 Values of

Vibrational temperature diagram derived from the HCN J = 32 lines given... | Download Scientific Diagram
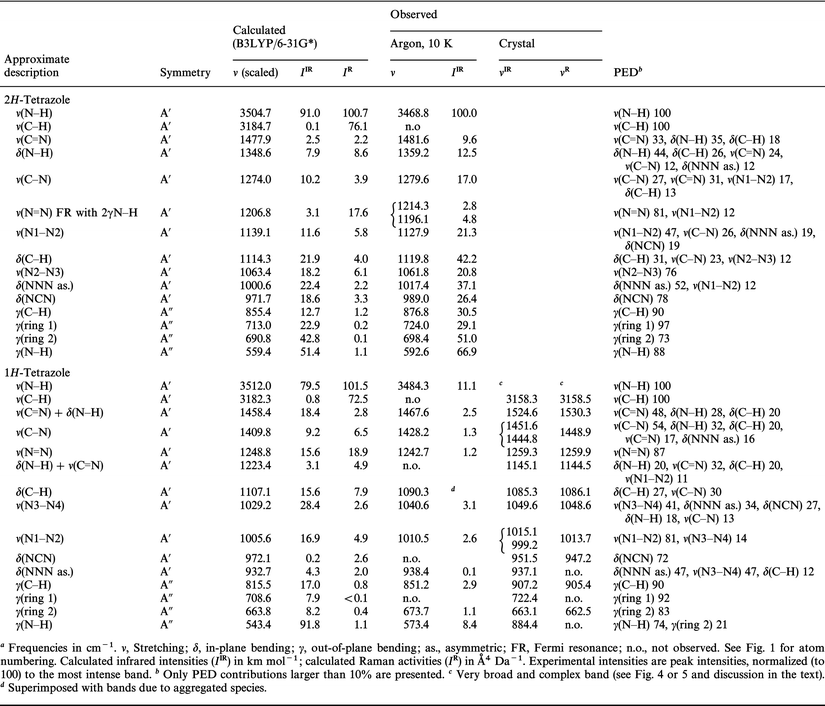
Low temperature matrix-isolation and solid state vibrational spectra of tetrazole - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/B103344C
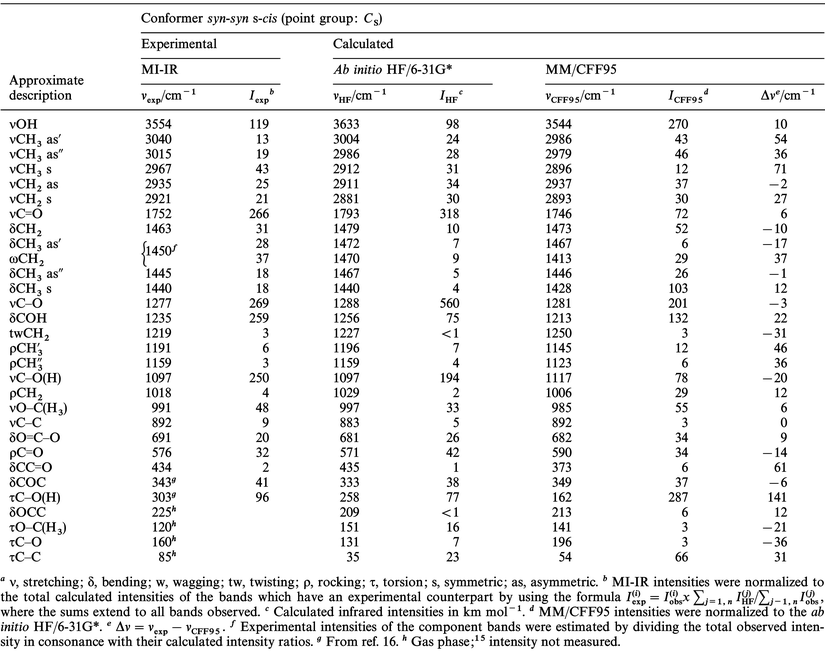
Structural and vibrational characterization of methyl glycolate in the low temperature crystalline and glassy states - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/A909291I

thermodynamics - At what temperature does the vibrational degree of freedom becomes significant for an ideal diatomic molecule? - Physics Stack Exchange