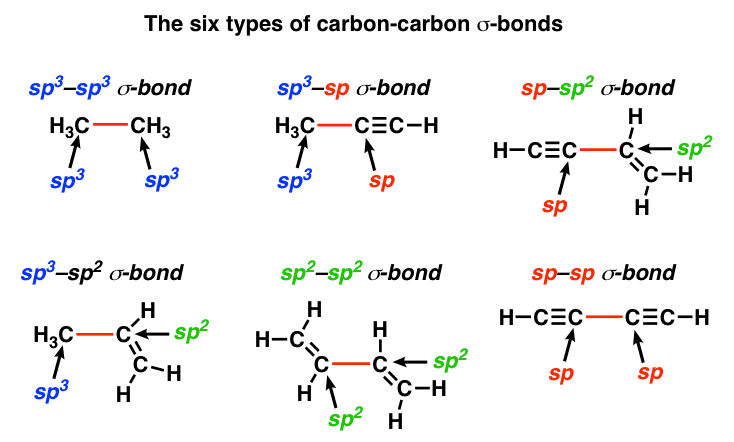
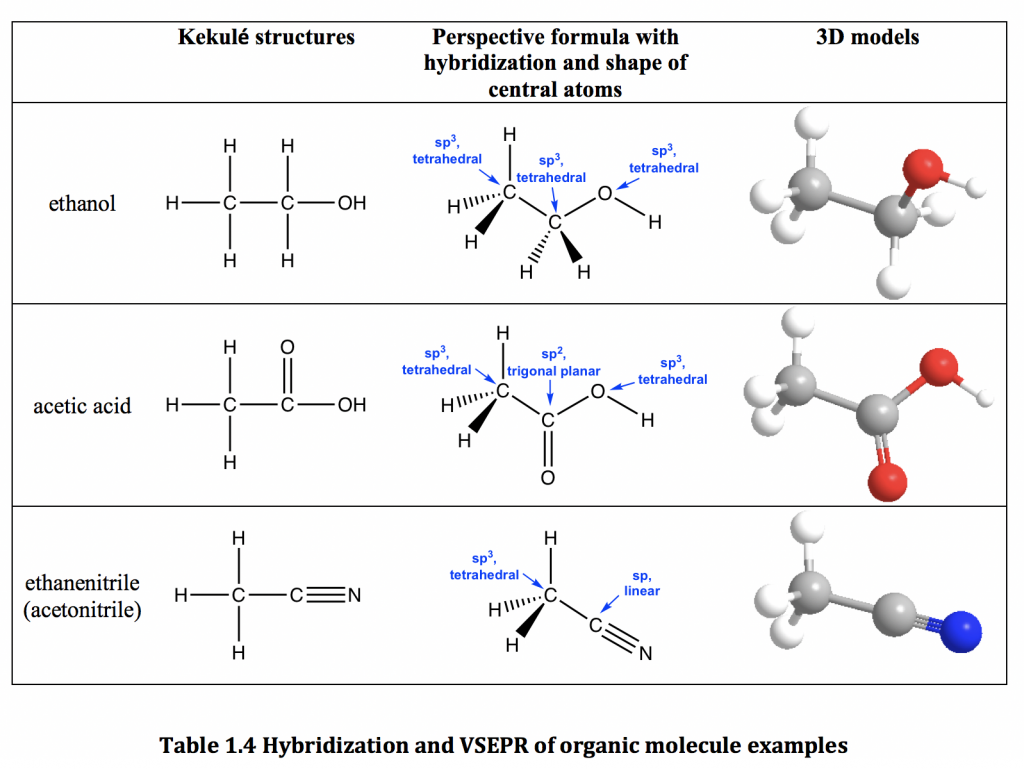
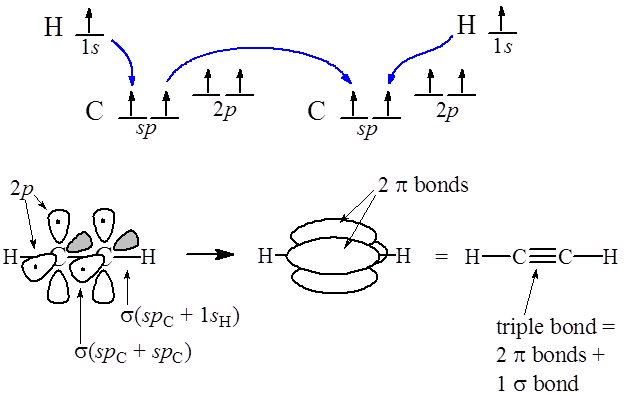
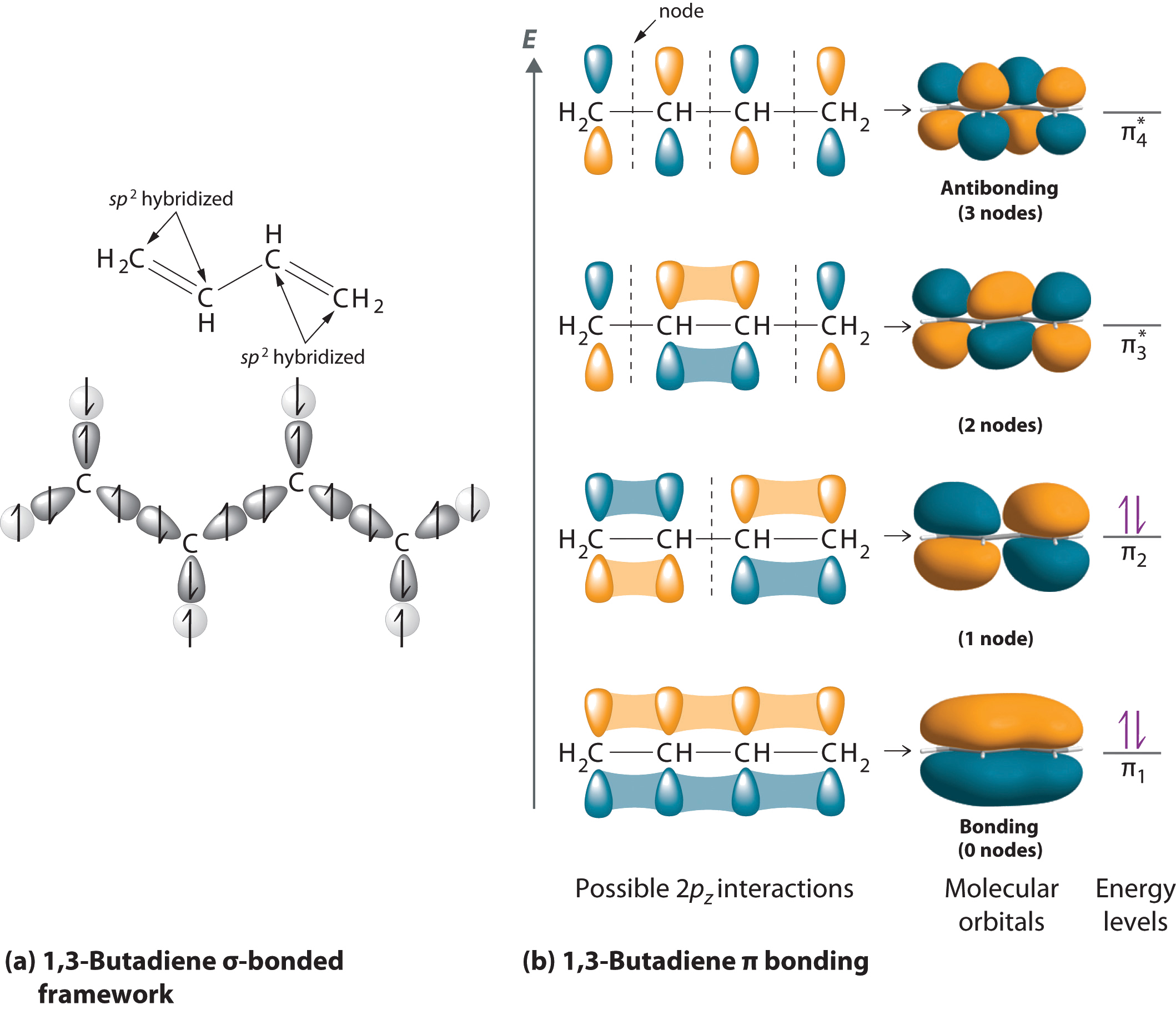
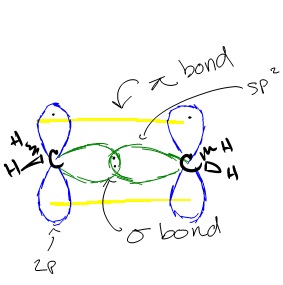
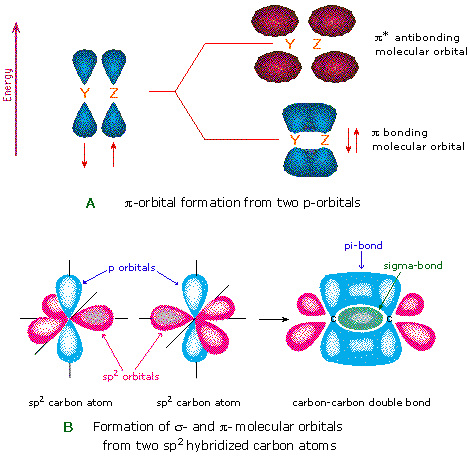
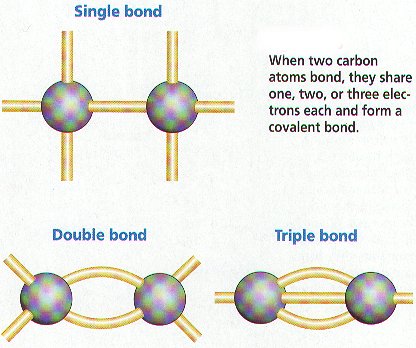
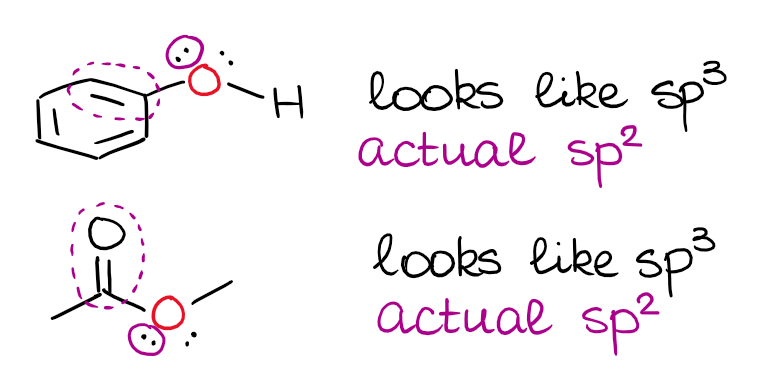Does sp2 hybridization mean that the atom will form three bonds (we're talking any atom, not just carbon)? - Quora

The different hybridization of carbon atom allows it to generate bonds... | Download Scientific Diagram

How does carbon hybridize its s and p orbitals and what effect does this have on the 3D structure and chemical behavior? Your answer must include diagrams of sp, sp2, and sp3