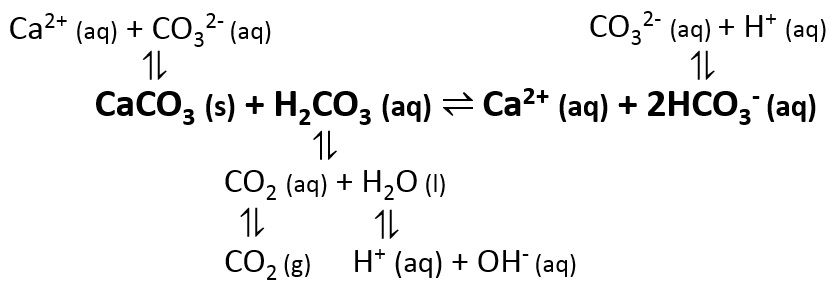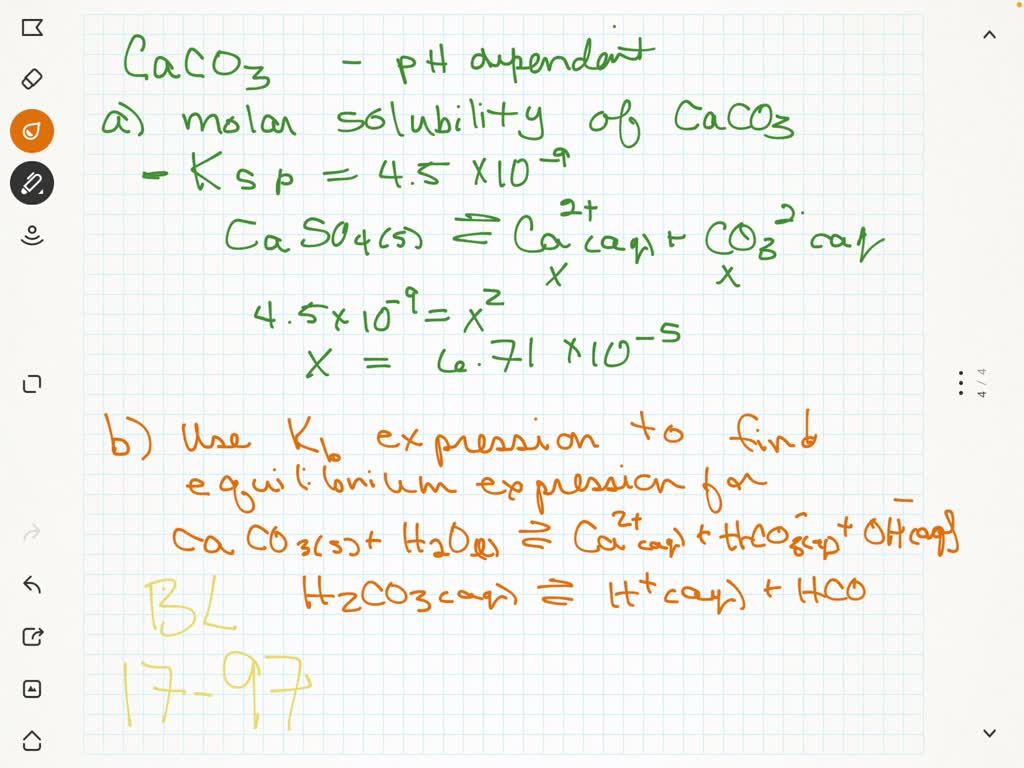
SOLVED: The KSP of CaCO3 is 3.369 × 10-9, the Ka of carbonic acid (H2CO3) is 4.400 × 10-7, the Ka of hydrogen carbonate ion (HCO3-) is 4.700 × 10-11, the Ka

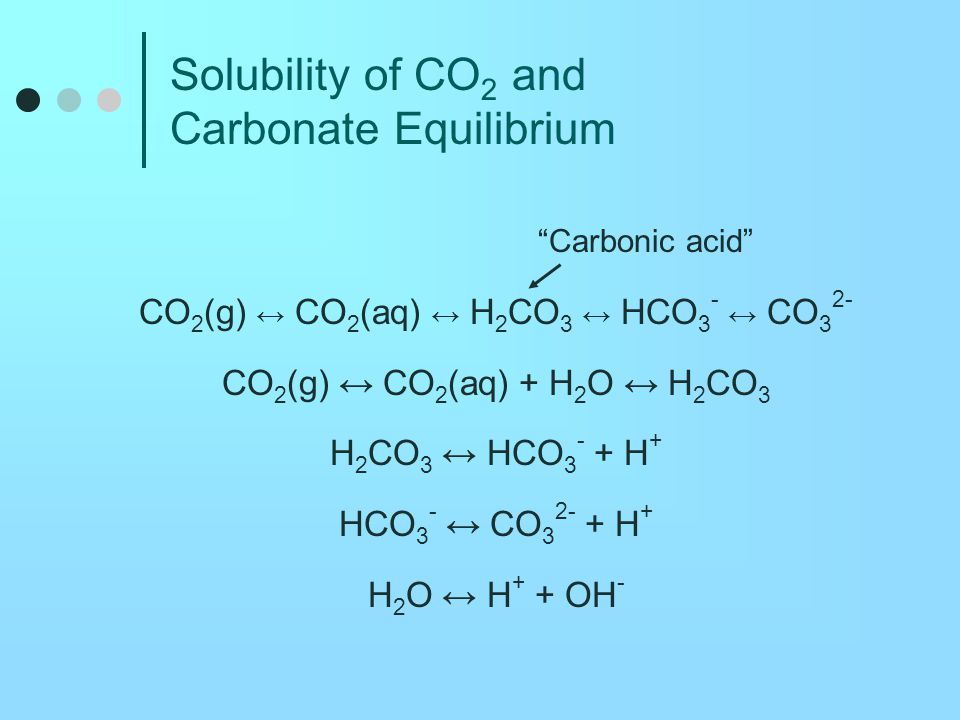
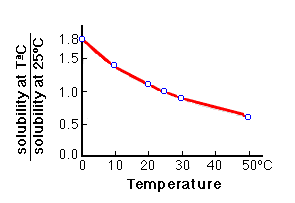
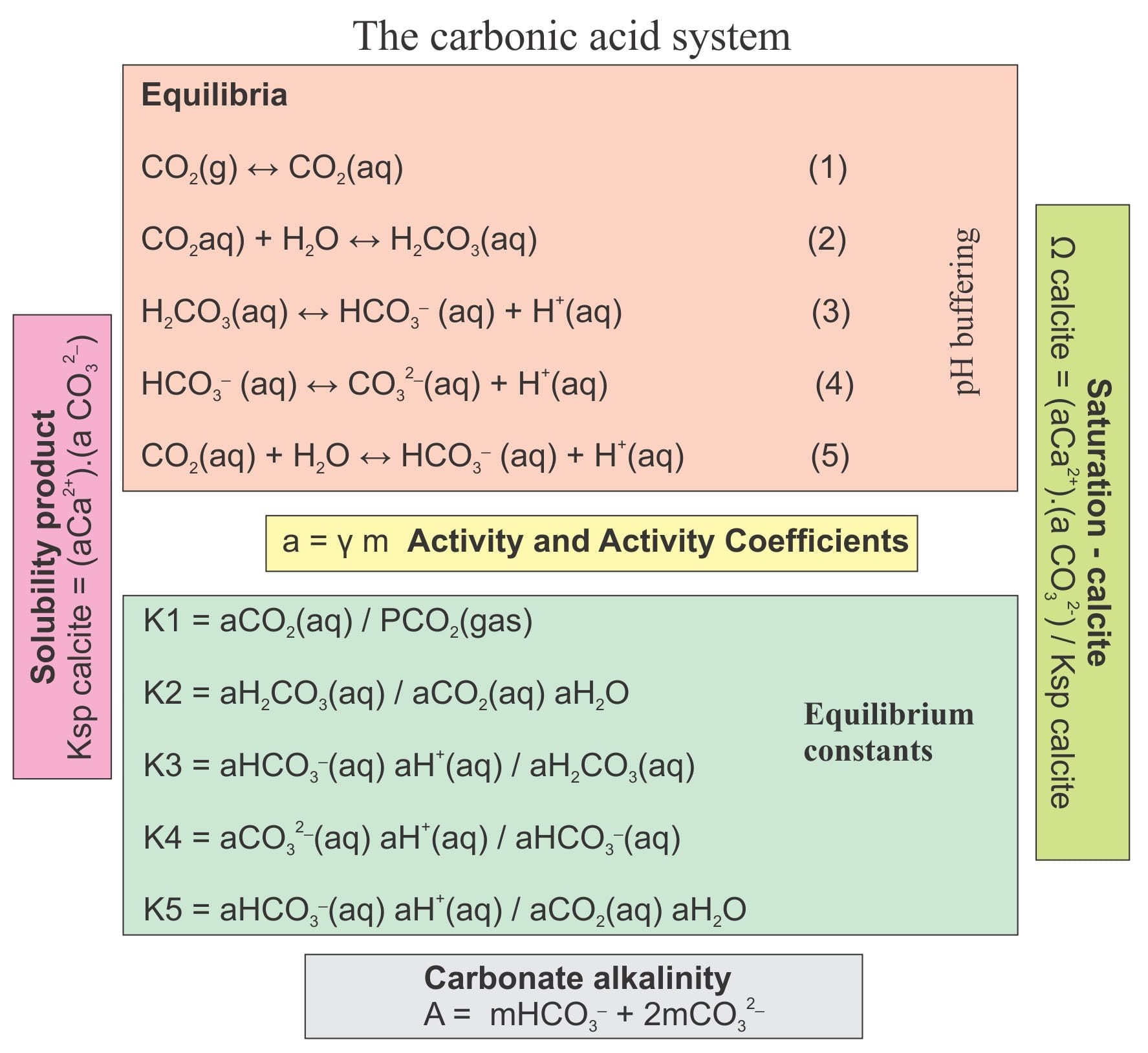
Summary of the reactions between carbon dioxide (CO2) with water (H2O)... | Download Scientific Diagram
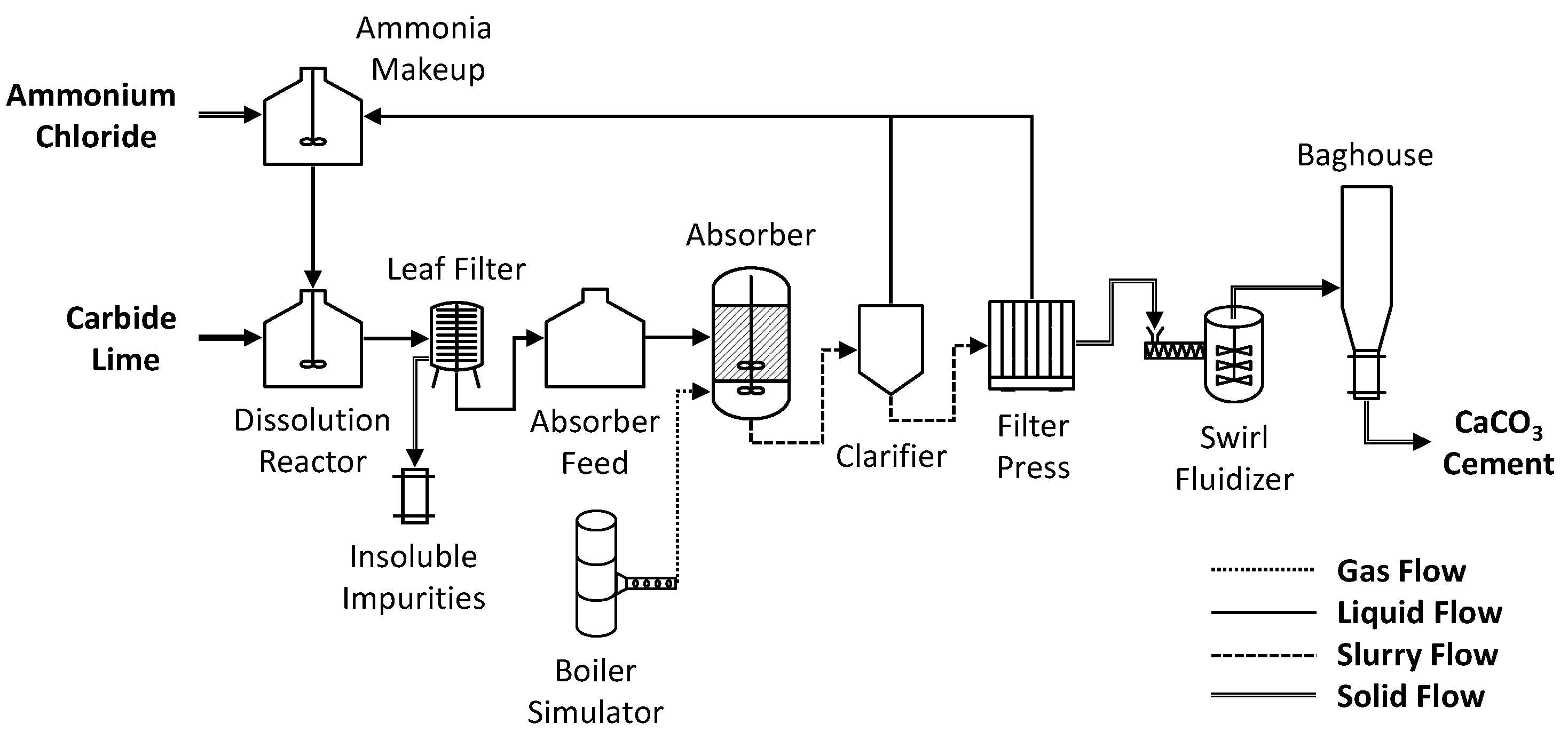
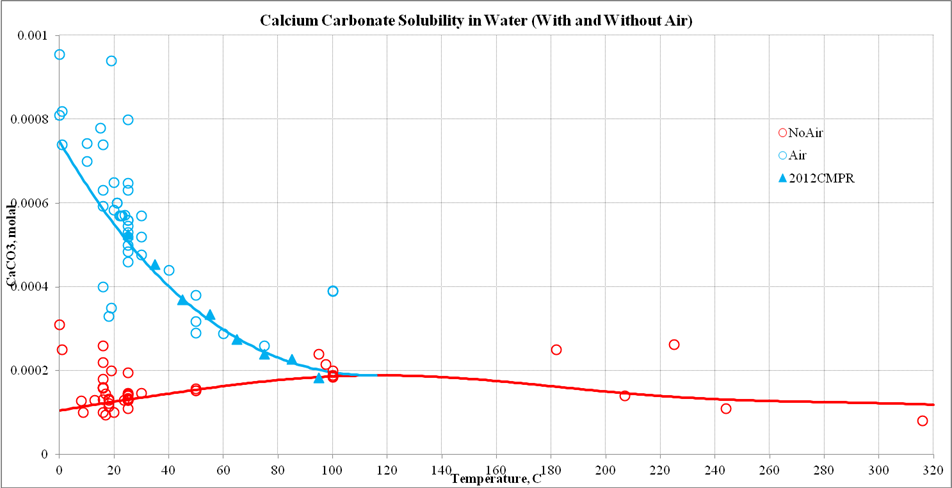
Materials | Free Full-Text | Calcium Carbonate Cement: A Carbon Capture, Utilization, and Storage (CCUS) Technique
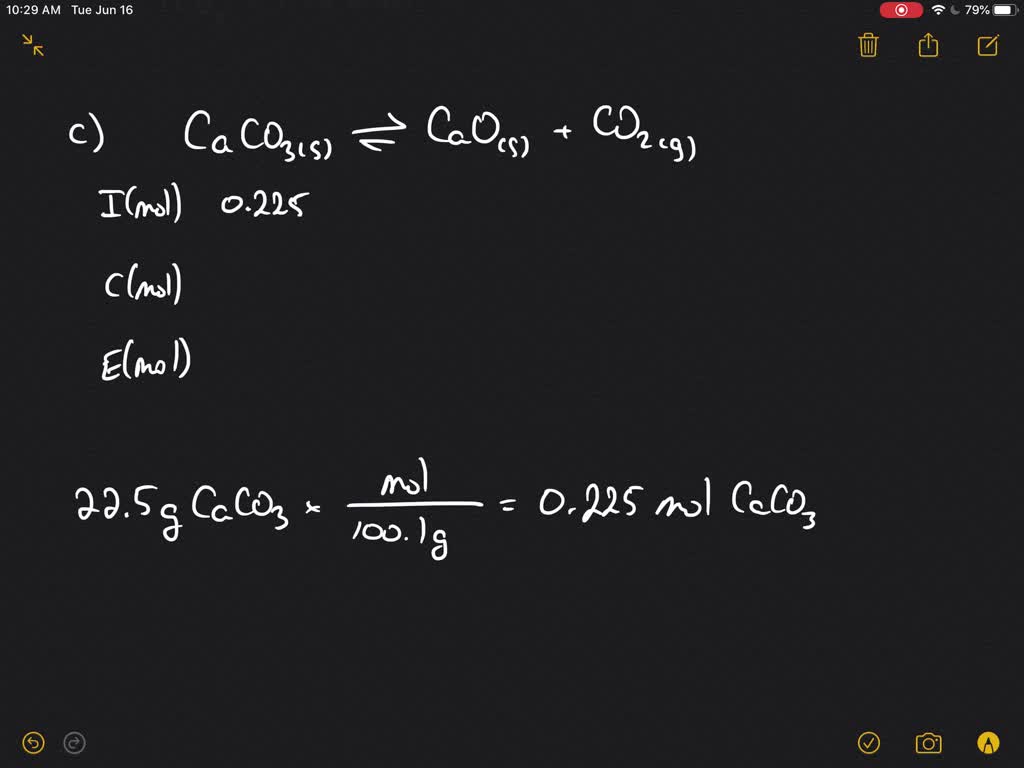
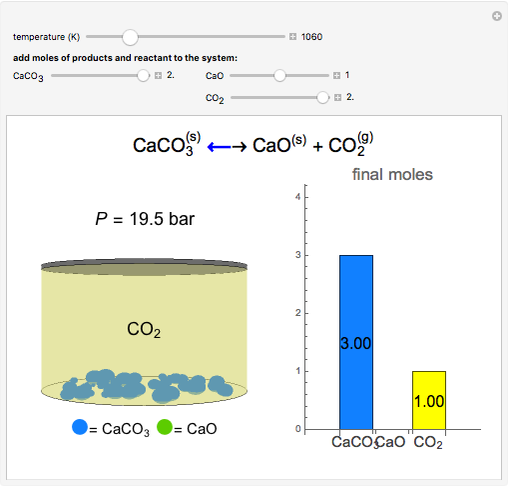
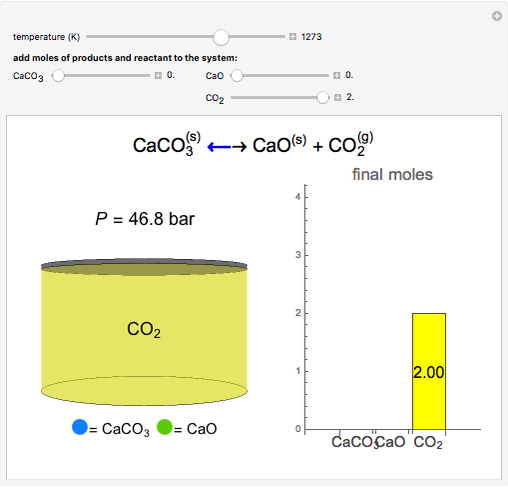
SOLVED:A The dissociation of calcium carbonate has an equilibrium constant of Kp=1.16 at 800^∘ C CaCO3(s) ⇄CaO(s)+CO2(g) (a) What is Kc for the reaction? (b) If you place 22.5 g of CaCO3
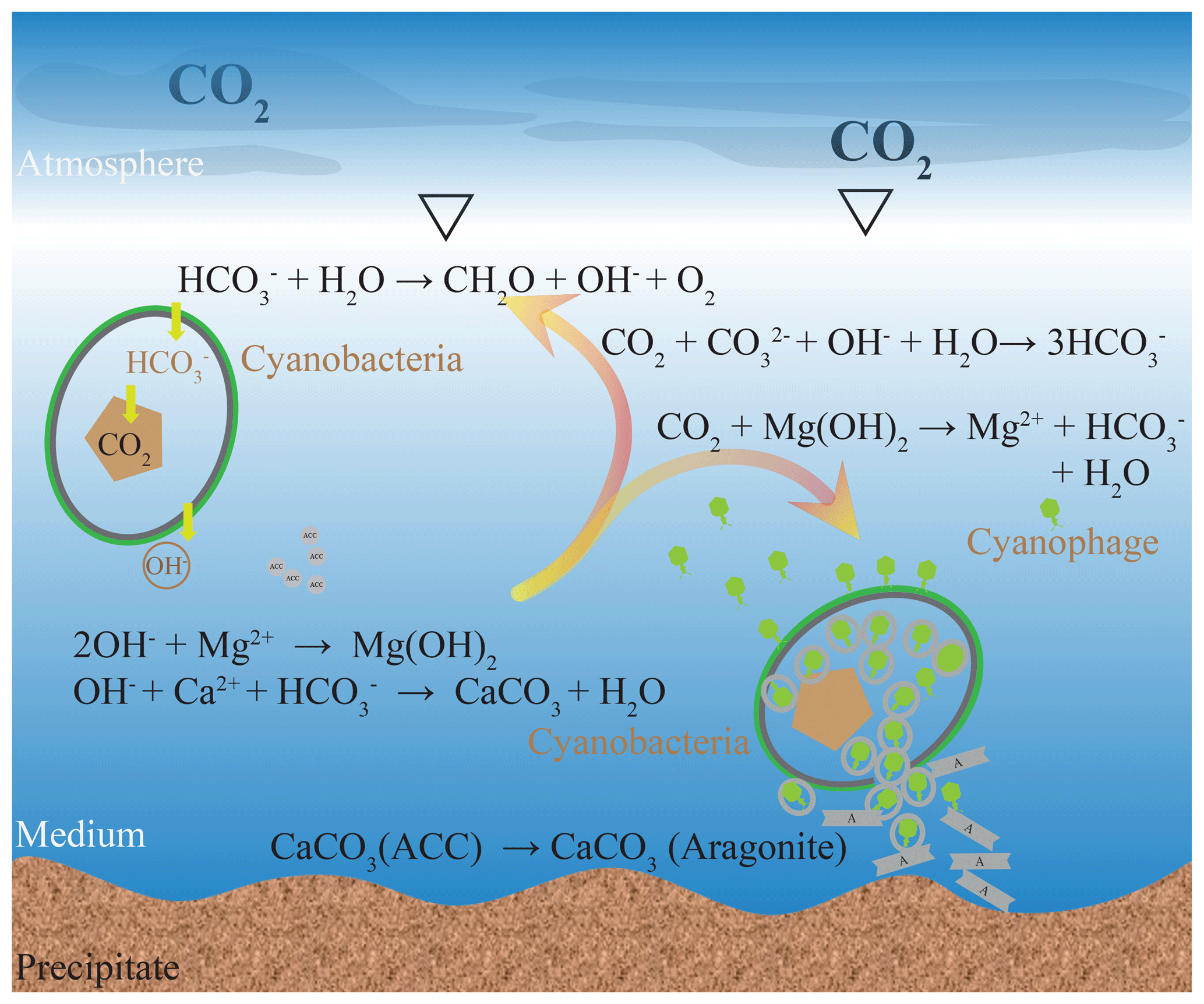
BG - Precipitation of calcium carbonate mineral induced by viral lysis of cyanobacteria: evidence from laboratory experiments

Ksp for calcium carbonate is 3 x 10^(-9). If you mix together 100 mL of 0.01 M calcium chloride with 100 mL of 10^(-5) M solution of potassium carbonate, will a precipitate form? | Homework.Study.com

Question Video: Determining the Expression for the Equilibrium Constant for the Decomposition of Calcium Carbonate | Nagwa