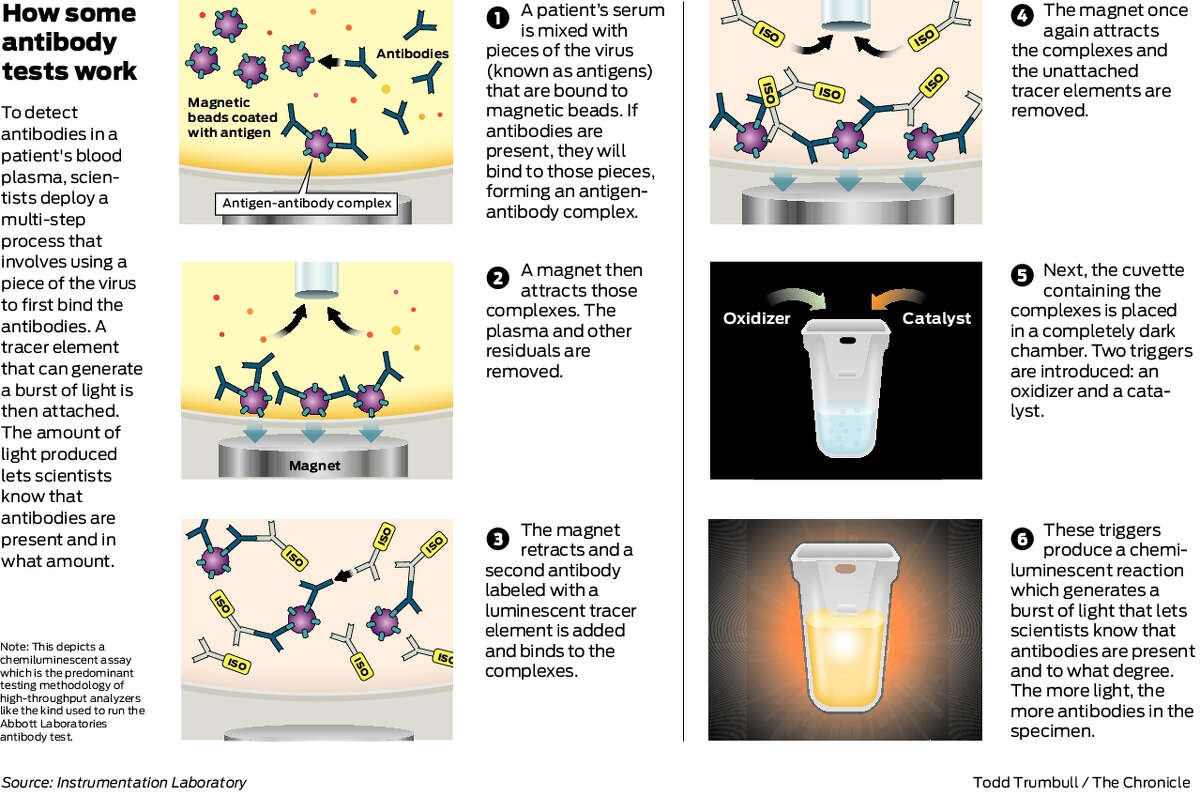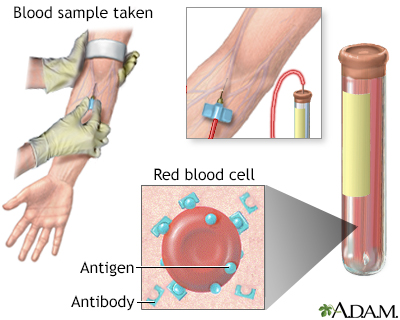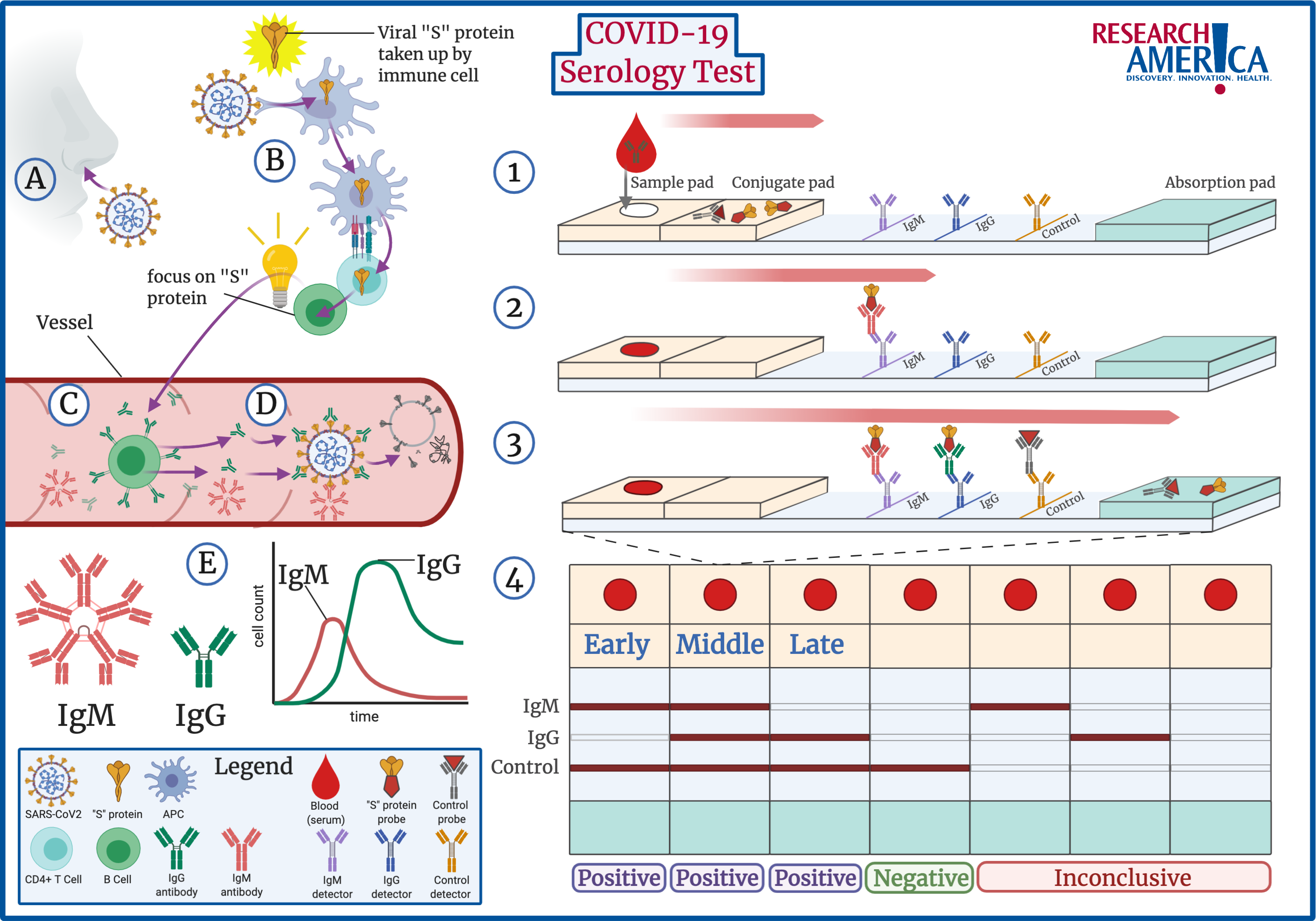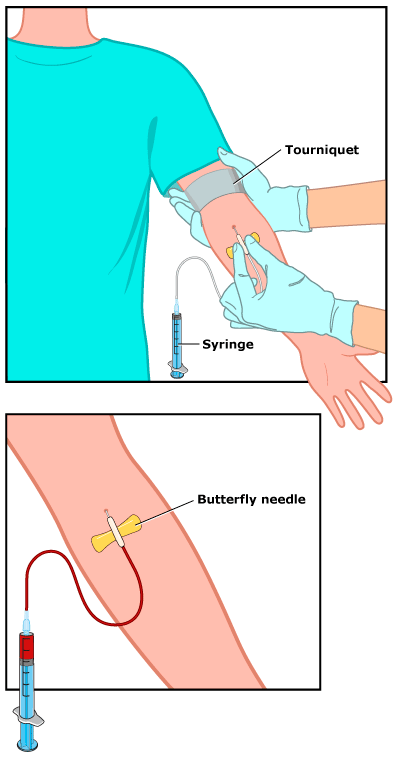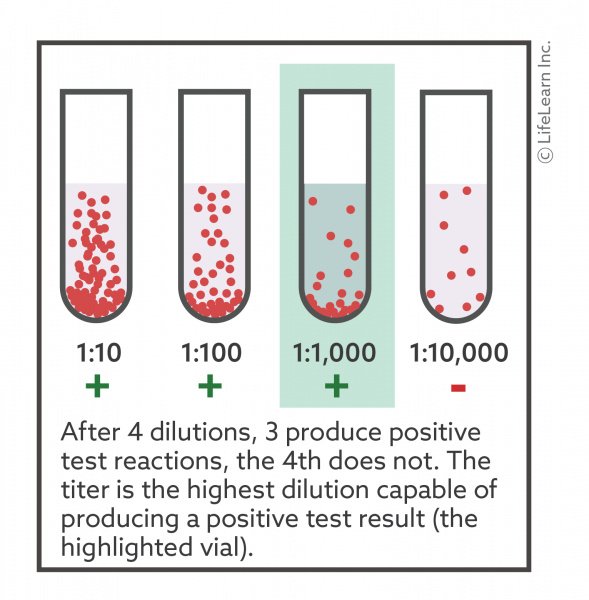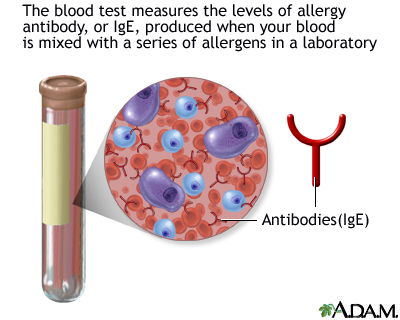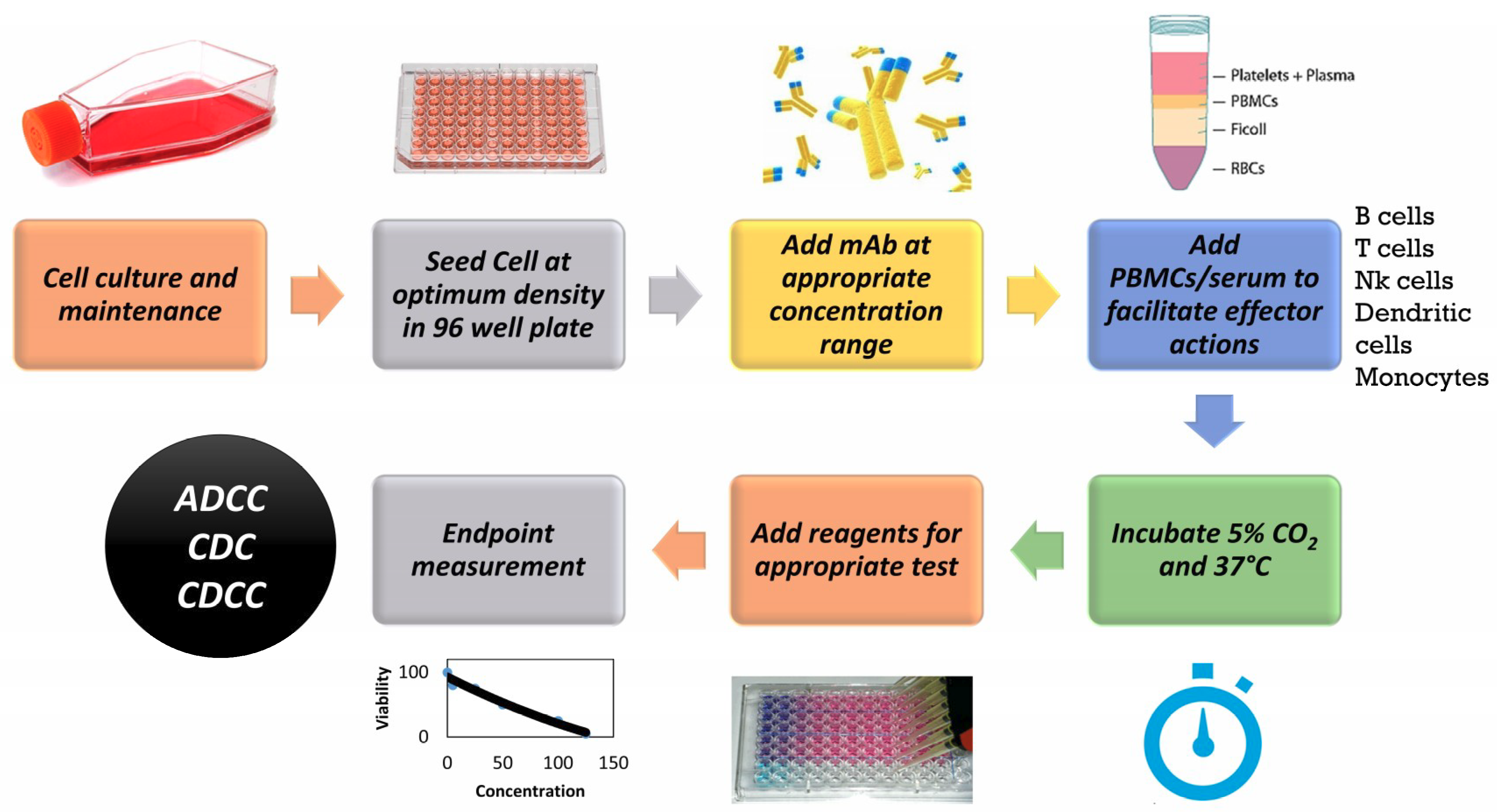
Antibodies | Free Full-Text | Applicability of Traditional In Vitro Toxicity Tests for Assessing Adverse Effects of Monoclonal Antibodies: A Case Study of Rituximab and Trastuzumab

Multiplex profiling of serum proteins in solution using barcoded antibody fragments and next generation sequencing | Communications Biology

Two new reference materials for the quality control of SARS-CoV-2 antibody tests released - Welcome!

Defining the features and duration of antibody responses to SARS-CoV-2 infection associated with disease severity and outcome | Science Immunology
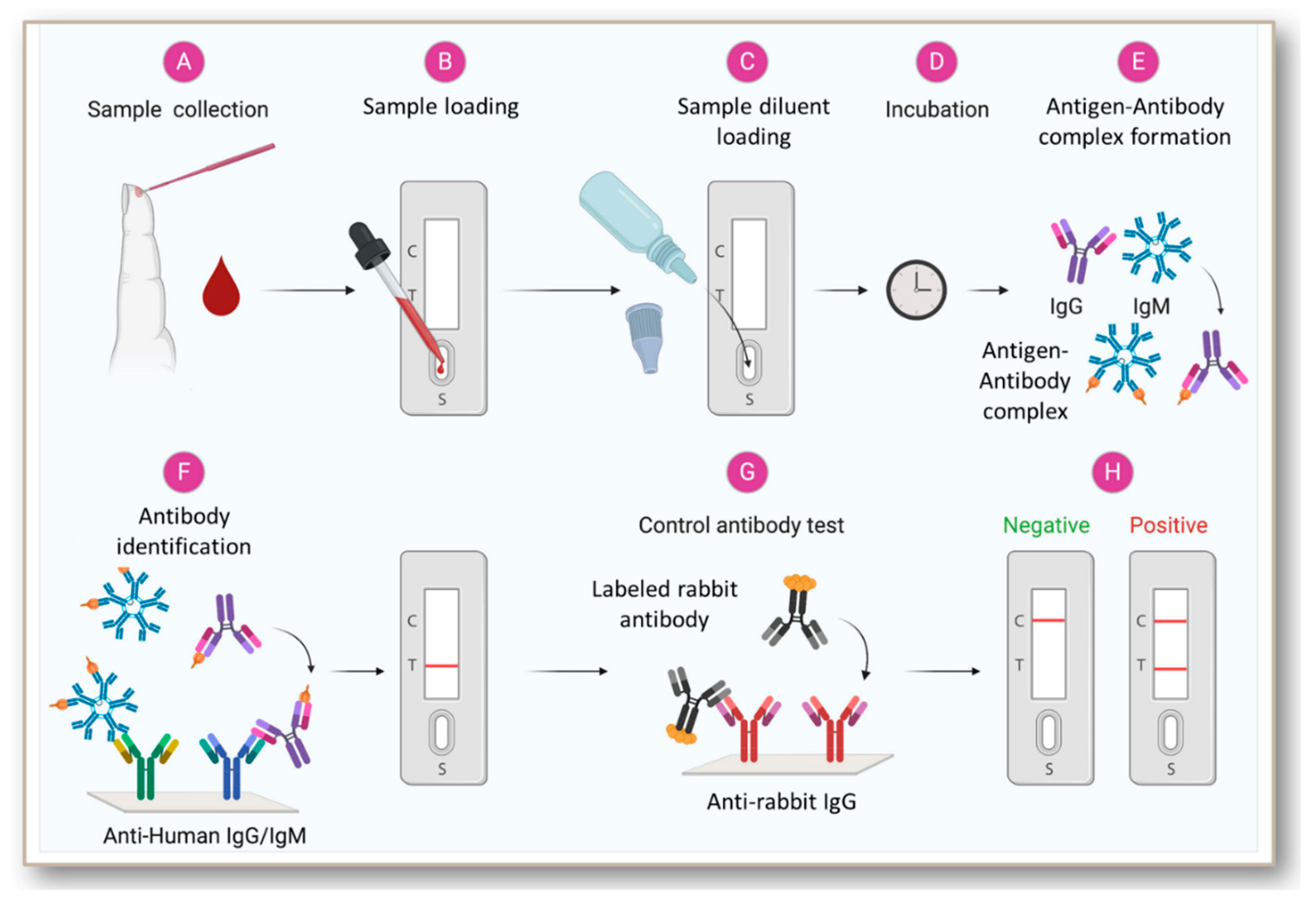
JCM | Free Full-Text | Rapid Antibody-Based COVID-19 Mass Surveillance: Relevance, Challenges, and Prospects in a Pandemic and Post-Pandemic World
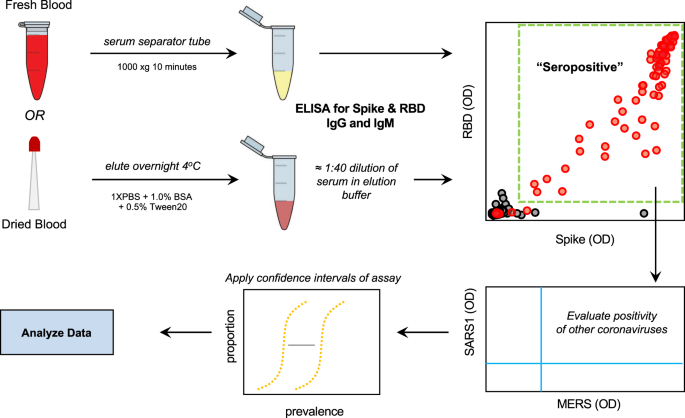
Standardization of ELISA protocols for serosurveys of the SARS-CoV-2 pandemic using clinical and at-home blood sampling | Nature Communications